Key Takeaways
-
Ubiquitous Challenge: Fouling — the unwanted buildup on surfaces — affects diverse fields from shipping and energy to healthcare and daily life.
-
Multifaceted Forces: It's driven by a complex interplay of physical (van der Waals, electrostatic), chemical (covalent, hydrogen, ionic), and biological forces.
-
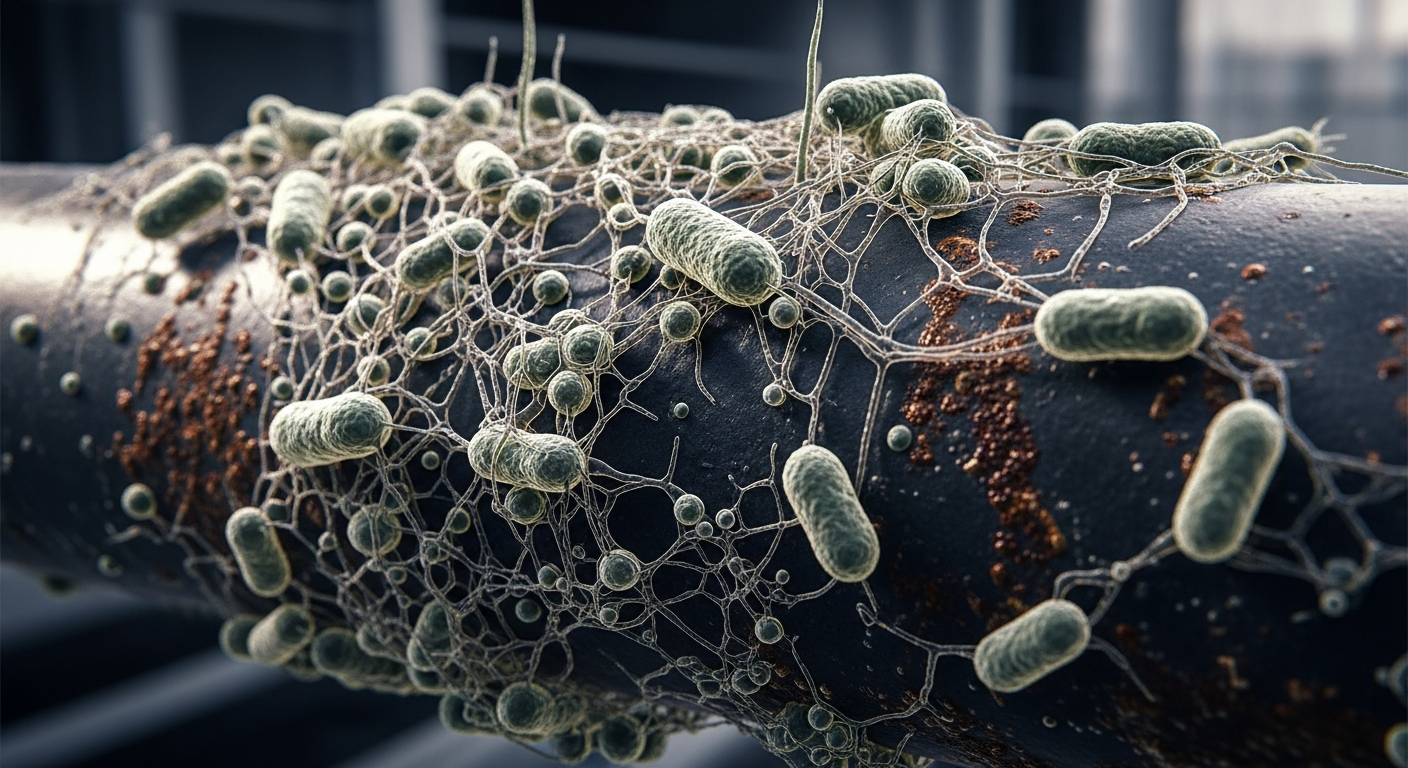
Microbial Influence: Microorganisms often initiate and accelerate fouling by forming protective biofilms, complicating prevention efforts.
-
Predictive Power: Mathematical modeling provides crucial insights, helping engineers and scientists anticipate and counteract fouling dynamics.
-
Innovation Imperative: Advanced fouling research is essential for developing more efficient, sustainable, and safer technologies worldwide.
The Unseen Adversary: An Introduction to Fouling
Imagine a world where surfaces remain perpetually pristine, unburdened by the relentless march of buildup. While a utopian vision, it highlights the pervasive nature of fouling – the accumulation of unwanted material on a surface. This phenomenon, seemingly mundane, is a sophisticated dance of interactions at the molecular and macroscopic levels, dictating efficiency, safety, and longevity across countless applications. From the gradual dulling of a water pipe to the critical degradation of a medical device, fouling is a complex adversary that demands a deep dive into its underlying scientific mechanisms.

First Contact: The Role of Physical Forces
Before chemical bonds can form or biological organisms can take hold, the initial approach of a foulant to a surface is governed by a suite of physical forces. These include the omnipresent van der Waals forces, the charge-driven electrostatic interactions, and the critical balance of hydrophobic interactions. Think of these as the 'first handshake' between a foulant and a surface. The topography of the surface – its roughness, its inherent charge, and how it repels or attracts water (wettability) – all play a pivotal role. A rough surface, for instance, offers more nooks and crannies for initial attachment, while a highly hydrophobic surface might effectively shrug off certain water-borne foulants.
The Molecular Embrace: Chemical Bonding
Once the foulant is in close proximity, the stage is set for chemical forces to take over, transforming transient contact into a more steadfast bond. Here, the molecular world comes alive with covalent bonds – the strongest, where atoms share electrons – alongside the more transient yet significant hydrogen bonds and ionic bonds. The nature and robustness of this 'molecular embrace' are deeply rooted in the chemical makeup of both the surface and the incoming foulant. Environmental factors, such as temperature and pH levels, act as orchestrators, influencing the strength and type of these crucial chemical interactions. This intricate bonding process often dictates the persistence and difficulty of removing a foulant.
Life's Sticky Gambit: The Microbial Dimension
In many of fouling's most persistent and problematic manifestations, life itself plays a central role. Microorganisms – bacteria, algae, fungi – are not just passengers but active participants, often initiating and exacerbating the fouling process. These microscopic architects produce extracellular polymeric substances (EPS), a sticky, protective matrix that not only cements them to the surface but also acts as a biological net, trapping other particles and organisms. The result is a complex, resilient community known as a biofilm. Understanding the 'microbial factor' is paramount, as these living layers can significantly amplify fouling's impact, from increased corrosion to reduced heat transfer efficiency and compromised public health.
Cracking the Code: Mathematical Modeling
To move beyond observation and towards prediction and control, researchers turn to the power of mathematics. Mathematical models are indispensable tools that capture the dynamic evolution of foulant accumulation and removal over time. Often expressed through differential equations, these models integrate factors such as mass transport (how foulants move to the surface), reaction kinetics (how quickly they attach), and the nuances of surface interactions. By solving these equations, scientists can simulate various scenarios, predicting how fouling systems will behave under different conditions. This predictive capability is vital, allowing for the strategic optimization of anti-fouling measures and the design of more resilient materials and systems. For instance, an equation like:
[ \frac{dM}{dt} = k \cdot C \cdot (1 - \theta) ]
describes the rate of foulant mass accumulation ($M$) over time ($t$), considering the mass transfer coefficient ($k$), foulant concentration ($C$), and existing surface coverage ($\theta$). Such models provide a framework to deconstruct and manipulate the intricate dance between foulants and surfaces.
Public Sentiment
-
"It's astounding how something as seemingly simple as a 'sticky surface' can have such profound economic and environmental implications. From cleaner energy systems to safer medical devices, understanding fouling feels incredibly important." – Anya Sharma, Materials Science Enthusiast
-
"In my line of work, fouling isn't just a nuisance; it's a constant battle that drives up maintenance costs and reduces operational lifespan. The advancements in this research are crucial for industrial efficiency." – Rajesh Kumar, Chemical Engineer
-
"The idea that microscopic life forms are behind some of the toughest fouling challenges is fascinating. It really underscores how intertwined biology is with everything, even our industrial infrastructure." – Dr. Lena Petrova, Microbiologist
Conclusion
Fouling research is a testament to the power of multidisciplinary science, weaving together engineering, biology, chemistry, and physics to tackle a pervasive global challenge. By systematically dissecting the physical, chemical, and biological forces at play, and by leveraging advanced mathematical modeling, scientists are steadily unraveling the complexities of this 'silent saboteur.' The insights gained from this relentless pursuit are not just academic victories; they are catalysts for real-world solutions, paving the way for innovations that promise to deliver more efficient energy systems, safer healthcare technologies, and a more sustainable future across our planet. The deeper we delve into the world of surface interactions, the more we empower ourselves to design a cleaner, more reliable world.